
Regulatory Advocacy
Federal agencies take the laws passed by Congress, interpret and implement them through the rulemaking process. The resulting regulations provide the requirements, standards, and processes needed to practically carry out laws. Regulatory advocacy refers to the efforts of organizations and individuals to influence government agencies as they engage in the rulemaking process.
Why Regulation matters
Federal agencies take Congress’s broad instructions and turn them into detailed rules that determine how a law is carried out. If a law is the diagnosis, regulation is the care plan.
Regulatory advocacy lets you shape that care plan. It’s one of the most direct, accessible ways to influence public policy. As part of the regulatory cycle, proposed regulations must open for public comment, and agencies must carefully consider each comment before issuing a final rule.
To follow an agency’s progress on a given regulation, the Office of Information and Regulatory Affairs (OIRA) tracks rules by agency and allows one to see where they are in the process. Proposed and final regulations, requests for information, and other notices are published in the Federal Register. Regulations open for comment are posted on Regulations.gov, where submitted comments are also posted for public review.
The Regulatory Cycle
While every agency has its quirks, the process follows a familiar rhythm:
- Drafting: Agencies research and write a proposed rule.
- Notice: They publish a Notice of Proposed Rulemaking (NPRM) in the Federal Register.
- Public Comment: A 30–60 day window opens for anyone to weigh in.
- Revision: Agencies review comments, make changes, and issue a final rule.
- Codification: The rule is added to the Code of Federal Regulations (CFR), the official record of all federal regulations.
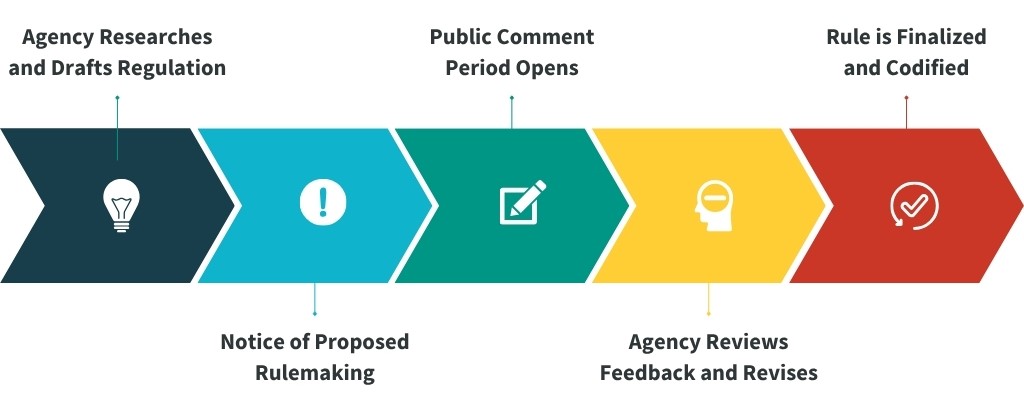
Once finalized, the rules get codified- in the Code of Federal Regulations (CFR), the official record of all federal regulations. Most health related rules live in Title 45 of the CFR. For more information please visit ANA’s Capitol Beat Blog, Regulation 101: How Nurses Can Shape Policy.
Regulatory Priorities
ANA’s regulatory advocacy encompasses all domains of the nursing profession and focuses on advocating for policies that promote wellbeing and safety for nurses and their patients. Our regulatory advocacy priorities are summed into five main topics:
For more information, check out ANA’s 2026 Regulatory and Policy Priorities.
ANA’s Work Across Executive Agencies
Federal agencies issue regulations and guidance for how laws are to be enforced, which impact many aspects of nursing practice.
Below lists the some of the Federal agencies and their relevant subagencies that issue policies that impact nursing practice, and the nursing-related topics of federal policies that fall under their domain or have influence over.
ANA Represents Nursing at CPT/RUC
The ANA is proud to be the only nursing association to have advisors at either the American Medical Association’s (AMA’s) CPT Editorial Committee or RVU Update Committee (RUC). While we work very closely with our partners at other nursing associations on any proposals that come out of these committees, ANA’s advisors represent the nursing profession before the AMA at these meetings.
 The CPT Editorial Panel creates new CPT codes or edits existing codes. These changes arise because either there is new technology or because procedures are now done differently than when the code was originally created resulting in an inaccurate CPT code descriptor. In some cases CPT codes can be deleted, but this generally only occurs when procedures are outdated and, therefore, are no longer being performed. Many applications for new or revised CPT codes come from medical specialty societies, such as ANA, but this is not a requirement. Device manufacturers can also submit applications and the CPT Editorial will give these applications the same thorough review that they give all applications.
The CPT Editorial Panel creates new CPT codes or edits existing codes. These changes arise because either there is new technology or because procedures are now done differently than when the code was originally created resulting in an inaccurate CPT code descriptor. In some cases CPT codes can be deleted, but this generally only occurs when procedures are outdated and, therefore, are no longer being performed. Many applications for new or revised CPT codes come from medical specialty societies, such as ANA, but this is not a requirement. Device manufacturers can also submit applications and the CPT Editorial will give these applications the same thorough review that they give all applications.
If the CPT Editorial Panel accepts the application and creates new codes, the next step in the process is going before the RUC. The RUC values the CPT codes and submits these values to CMS for the annual Physician Fee Schedule (PFS) rulemaking. ANA not only has advisors to represent ANA’s interests before the panel, but also has a seat on the RUC’s Practice Expense Subcommittee. This subcommittee reviews all of the applications and determines what medical supplies and devices should be used for the procedure and these items are then included in the final valuations submitted to CMS. CMS is not obligated to accept the RUC’s recommendations, but the vast majority of the RUC’s submissions are accepted and published in CMS’ PFS rule.
ANA staff works closely with our volunteer advisors preparing them for the multiple meetings held each year by both the CPT Editorial Committee and the RUC. The input provided by our advisors, along with the various committee recommendations, then inform ANA’s comments on the reimbursement sections of the PFS proposed rule.
RUC Survey
If the procedures are performed, or expected to be performed, by nurses, ANA will then send out surveys to a random sampling of members. This sampling is based on members self-identifying what their specialty is and the information gathered is used only for the stated RUC purpose of proper reimbursement.
If you receive these surveys, ANA implores the recipient, if they perform the procedure, to fill the survey out as accurately as possible. Although the surveys may span multiple specialties, including the nursing perspective is vital to maintaining nursing representation in the valuation process. ANA recognizes that completing these surveys, particularly those involving numerous codes, can be time-intensive; however, participation is a key to ensure nurses’ voices are heard.
